Khác biệt giữa bản sửa đổi của “Sinh khả dụng”
Không có tóm lược sửa đổi |
|||
| Dòng 1: | Dòng 1: | ||
| ⚫ | Trong [[Dược học]], '''Sinh khả dụng''' (hay như trong tiếng Anh là '''bioavailability''' (BA)) là đại lượng chỉ tốc độ và mức độ hấp thu dược chất từ một chế phẩm bào chế và tuần hoàn chung một cách nguyên vẹn và đưa đến nơi tác dụng, từ đó tiếp tục được chuyển hoá và thải hồi. Từ định nghĩa, thuốc theo đường tiêm tĩnh mạch có sinh khả dụng là 100%.<ref>Griffin, J.P. The Textbook of Pharmaceutical Medicine (6th Ed.). New Jersey: BMJ Books. ISBN 978-1-4051-8035-1{{pn|date=February 2013}}</ref> Tuy vậy, khi thuốc được dùng bằng các cách thức khác nhau (như đường uống) thì sinh khả dụng của thuốc{{cref|TH}} thường giảm (do hấp thu không hoàn toàn và các giai đoạn đầu của quá trình trao đỗi chất) hay thay đỗi tuỳ thuộc thể trạng bệnh nhân. Sinh khả dụng được xem là một công cụ thiết yếu trong sinh dược học, đây là đại lượng quan trọng để xác định và tính toán liều dùng cho các dạng bào chế không theo đường tĩnh mạch. |
||
For [[dietary supplements]], herbs and other nutrients in which the route of administration is nearly always oral, bioavailability generally designates simply the quantity or fraction of the ingested dose that is absorbed.<ref>{{cite journal |first1=Robert P. |last1=Heaney |title=Factors Influencing the Measurement of Bioavailability, Taking Calcium as a Model |journal=The Journal of Nutrition |pmid=11285351 |url=http://jn.nutrition.org/cgi/pmidlookup?view=long&pmid=11285351 |year=2001 |volume=131 |issue=4 |pages=1344S–8S}}</ref> |
|||
Bioavailability is defined slightly differently for drugs as opposed to [[dietary supplements]] primarily due to the method of administration and [[Food and Drug Administration]] regulations. |
|||
Bioaccessibility is a concept related to bioavailability in the context of [[biodegradation]] and [[environmental pollution]]. A [[molecule]] (often a [[persistent organic pollutant]]) is said to be bioavailable when ''"[it] is available to cross an organism’s [[cellular membrane]] from the environment, if the organism has access to the chemical."''<ref>{{cite journal |doi=10.1021/es040548w |title=Peer Reviewed: Defining Bioavailability and Bioaccessibility of Contaminated Soil and Sediment is Complicated |year=2004 |last1=Semple |first1=Kirk. T. |last2=Doick |first2=Kieron J. |last3=Jones |first3=Kevin C. |last4=Burauel |first4=Peter |last5=Craven |first5=Andrew |last6=Harms |first6=Hauke |journal=Environmental Science & Technology |volume=38 |issue=12 |pages=228A–31A}}</ref> |
|||
| ⚫ | |||
===Trong Dược học=== |
|||
Trong Dược học, Sinh khả dụng là một địa lượng chỉ tốc độ và mức độ hấp thu dược chất từ một chế phầm bào chế vào tuần hoàn chung một cách nguyên vẹn và đưa đến nơi tác dụng.<ref>Shargel, L.; Yu, A.B. (1999). ''Applied biopharmaceutics & pharmacokinetics'' (4th ed.). New York: McGraw-Hill. ISBN 0-8385-0278-4{{pn|date=February 2013}}</ref> |
Trong Dược học, Sinh khả dụng là một địa lượng chỉ tốc độ và mức độ hấp thu dược chất từ một chế phầm bào chế vào tuần hoàn chung một cách nguyên vẹn và đưa đến nơi tác dụng.<ref>Shargel, L.; Yu, A.B. (1999). ''Applied biopharmaceutics & pharmacokinetics'' (4th ed.). New York: McGraw-Hill. ISBN 0-8385-0278-4{{pn|date=February 2013}}</ref> |
||
Được ký hiệu bằng chữ ''f'' (hay nếu ở dạng phần trăm là ''F''). |
Được ký hiệu bằng chữ ''f'' (hay nếu ở dạng phần trăm là ''F''). |
||
| ⚫ | |||
=== Trong Khoa học Dinh dưỡng === |
|||
| ⚫ | Trong [[Dược học]], '''Sinh khả dụng''' (hay như trong tiếng Anh là '''bioavailability''' (BA)) là đại lượng chỉ tốc độ và mức độ hấp thu dược chất từ một chế phẩm bào chế và tuần hoàn chung một cách nguyên vẹn và đưa đến nơi tác dụng, từ đó tiếp tục được chuyển hoá và thải hồi. Từ định nghĩa, thuốc theo đường tiêm tĩnh mạch có sinh khả dụng là 100%.<ref>Griffin, J.P. The Textbook of Pharmaceutical Medicine (6th Ed.). New Jersey: BMJ Books. ISBN 978-1-4051-8035-1{{pn|date=February 2013}}</ref> Tuy vậy, khi thuốc được dùng bằng các cách thức khác nhau (như đường uống) thì sinh khả dụng của thuốc{{cref|TH}} thường giảm (do hấp thu không hoàn toàn và các giai đoạn đầu của quá trình trao đỗi chất) hay thay đỗi tuỳ thuộc thể trạng bệnh nhân. Sinh khả dụng được xem là một công cụ thiết yếu trong sinh dược học, đây là đại lượng quan trọng để xác định và tính toán liều dùng cho các dạng bào chế không theo đường tĩnh mạch. |
||
In nutritional sciences, which covers the intake of nutrients and non-drug dietary ingredients, the concept of bioavailability lacks the well-defined standards associated with the pharmaceutical industry. The pharmacological definition cannot apply to these substances because utilization and absorption is a function of the nutritional status and physiological state of the subject,<ref>{{cite journal |pmid=11285351 |year=2001 |last1=Heaney |first1=Robert P. |title=Factors Influencing the Measurement of Bioavailability, Taking Calcium as a Model |volume=131 |issue=4 Suppl |pages=1344S–8S |journal=The Journal of Nutrition |url=http://jn.nutrition.org/cgi/pmidlookup?view=long&pmid=11285351}}</ref> resulting in even greater differences from individual to individual (inter-individual variation). Therefore, bioavailability for dietary supplements can be defined as the proportion of the administered substance capable of being absorbed and available for use or storage.<ref name="Srinivasan">{{cite journal |pmid=11285352 |year=2001 |last1=Srinivasan |first1=V. Srini |title=Bioavailability of Nutrients: A Practical Approach to In Vitro Demonstration of the Availability of Nutrients in Multivitamin-Mineral Combination Products |volume=131 |issue=4 Suppl |pages=1349S–50S |journal=The Journal of Nutrition |url=http://jn.nutrition.org/cgi/pmidlookup?view=long&pmid=11285352}}</ref> |
|||
In both pharmacology and nutrition sciences, bioavailability is measured by calculating the [[area under curve]] (AUC) of the drug concentration time profile. |
|||
=== Trong Khoa học Môi trường=== |
|||
Bioavailability is commonly a limiting factor in the production of crops (due to solubility limitation or adsorption of plant nutrients to soil colloids) and in the removal of toxic substances from the food chain by microorganisms (due to sorption to or partitioning of otherwise degradable substances into inaccessible phases in the environment). A noteworthy example for agriculture is plant phosphorus deficiency induced by precipitation with iron and aluminum phosphates at low soil pH and precipitation with calcium phosphates at high soil pH.<ref>{{cite journal |doi=10.1023/A:1013351617532 |year=2001 |last1=Hinsinger |first1=Philippe |title=Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review |journal=Plant and Soil |volume=237 |issue=2 |pages=173–95}}</ref> Toxic materials in soil, such as lead from paint may be rendered unavailable to animals ingesting contaminated soil by supplying phosphorus fertilizers in excess.<ref>{{cite journal |doi=10.1021/es00046a007 |title=In situ lead immobilization by apatite |year=1993 |last1=Ma |first1=Qi Ying |last2=Traina |first2=Samuel J. |last3=Logan |first3=Terry J. |last4=Ryan |first4=James A. |journal=Environmental Science & Technology |volume=27 |issue=9 |pages=1803–10}}</ref> Organic pollutants such as solvents or pesticides may be rendered unavailable to microorganisms and thus persist in the environment when they are adsorbed to soil minerals<ref>{{cite journal |doi=10.1002/etc.5620190904 |title=Effects of sorption on the biodegradation of 2-methylpyridine in aqueous suspensions of reference clay minerals |year=2000 |last1=O'Loughlin |first1=Edward J. |last2=Traina |first2=Samuel J. |last3=Sims |first3=Gerald K. |journal=Environmental Toxicology and Chemistry |volume=19 |issue=9 |pages=2168–74}}</ref> or partition into hydrophobic organic matter.<ref>{{cite journal |doi=10.1002/(SICI)1096-9063(199905)55:5<598::AID-PS962>3.0.CO;2-N |title=Factors controlling degradation of pesticides in soil |year=1999 |last1=Sims |first1=Gerald K |last2=Cupples |first2=Alison M |journal=Pesticide Science |volume=55 |issue=5 |pages=598–601}}</ref> |
|||
== Sinh khả dụng tuyệt đối == |
== Sinh khả dụng tuyệt đối == |
||
| Dòng 32: | Dòng 15: | ||
: <math>F_{abs} = 100 \cdot \frac{AUC_{po} \cdot D_{iv}}{AUC_{iv} \cdot D_{po}}</math> |
: <math>F_{abs} = 100 \cdot \frac{AUC_{po} \cdot D_{iv}}{AUC_{iv} \cdot D_{po}}</math> |
||
Therefore, a drug given by the intravenous route will have an absolute bioavailability of 100% (''f''=1), whereas drugs given by other routes usually have an absolute bioavailability of less than one. |
|||
If we compare the two different dosage forms having same active ingredients and compare the two drug bioavailability is called comparative bioavailability. |
|||
Although knowing the true extent of systemic absorption (referred to as absolute bioavailability) is clearly useful, in practice it is not determined as frequently as one may think. The reason for this is that its assessment requires an intravenous reference, that is, a route of administration that guarantees that all of the administered drug reaches the systemic circulation. Such studies come at considerable cost, not least of which is the necessity to conduct preclinical toxicity tests to ensure adequate safety, as well as there being potential problems due to solubility limitations. These limitations may be overcome, however, by administering a very low dose (typically a few micrograms) of an isotopically labelled drug concomitantly with a therapeutic non-labelled oral dose. Providing the isotopically-labelled intravenous dose is sufficiently low so as not to perturb the systemic drug concentrations achieved from the absorbed oral dose, then the intravenous and oral pharmacokinetics can be deconvoluted by virtue of the their different isotopic constitution and thereby determine the oral and intravenous pharmacokinetics from the same dose administration. This technique eliminates pharmacokinetic issues on non-equivalent clearance as well as enabling the intravenous dose to be administered with a minimum of toxicology and formulation. The technique was first applied using stable-isotopes such as C-13 and mass-spectrometry to distinguish the isotopes by mass difference. More recently, C-14 labelled drugs are administered intravenously and accelerator mass spectrometry (AMS) used to measure the isotopically labelled drug along with mass spectrometry for the unlabelled drug.<ref>{{cite journal |doi=10.1517/17425255.2.3.419 |title=The use of isotopes in the determination of absolute bioavailability of drugs in humans |year=2006 |last1=Lappin |first1=Graham |last2=Rowland |first2=Malcolm |last3=Garner |first3=R Colin |journal=Expert Opinion on Drug Metabolism & Toxicology |volume=2 |issue=3 |pages=419–27 |pmid=16863443}}</ref> |
|||
There is no regulatory requirement to define the intravenous pharmacokinetics or absolute bioavailability however regulatory authorities do sometimes ask for absolute bioavailbility information of the extravascular route in cases in which the bioavailability is apparently low or variable and there is a proven relationship between the [[pharmacodynamics]] and the pharmacokinetics at therapeutic doses. In all such cases, to conduct an absolute bioavailability study requires that the drug be given intravenously.<ref name="Graham Lappin 2008">{{cite journal |doi=10.1517/17425255.4.8.1021 |title=Biomedical accelerator mass spectrometry: Recent applications in metabolism and pharmacokinetics |year=2008 |last1=Lappin |first1=Graham |last2=Stevens |first2=Lloyd |journal=Expert Opinion on Drug Metabolism & Toxicology |volume=4 |issue=8 |pages=1021–33 |pmid=18680438}}</ref> |
|||
Intravenous administration of a developmental drug can provide valuable information on the fundamental pharmacokinetic parameters of [[volume of distribution]] (V) and [[Clearance (medicine)|clearance]] (CL).<ref name="Graham Lappin 2008" /> |
|||
== Sinh khả dụng tương đối và tương đương sinh học == |
|||
In pharmacology, relative bioavailability measures the bioavailability (estimated as the AUC) of a formulation (''A'') of a certain drug when compared with another formulation (''B'') of the same drug, usually an established standard, or through administration via a different route. When the standard consists of intravenously administered drug, this is known as absolute bioavailability (see [[#Absolute bioavailability|above]]). |
|||
: <math>F_{rel} = 100 \cdot \frac{AUC_A \cdot D_B}{AUC_B \cdot D_A}</math> |
|||
Relative bioavailability is one of the measures used to assess [[bioequivalence]] (BE) between two drug products. For FDA approval, a generic manufacturer must demonstrate that the 90% [[confidence interval]] for the ratio of the mean responses (usually of AUC and the maximum concentration, C<sub>max</sub>) of its product to that of the "Brand Name drug"{{cref|OB}} is within the limits of 80% to 125%. While AUC refers to the extent of bioavailability, C<sub>max</sub> refers to the rate of bioavailability. When T<sub>max</sub> is given, it refers to the time it takes for a drug to reach C<sub>max</sub>. |
|||
While the mechanisms by which a formulation affects bioavailability and bioequivalence have been extensively studied in drugs, formulation factors that influence bioavailability and bioequivalence in nutritional supplements are largely unknown.<ref>{{cite journal |pmid=11285360 |year=2001 |last1=Hoag |first1=Stephen W. |last2=Hussain |first2=Ajaz S. |title=The Impact of Formulation on Bioavailability: Summary of Workshop Discussion |volume=131 |issue=4 Suppl |pages=1389S–91S |journal=The Journal of Nutrition |url=http://jn.nutrition.org/cgi/pmidlookup?view=long&pmid=11285360}}</ref> As a result, in nutritional sciences, relative bioavailability or bioequivalence is the most common measure of bioavailability, comparing the bioavailability of one formulation of the same dietary ingredient to another. |
|||
== Các nhân tố ảnh hưởng dến sinh khả dụng == |
|||
The absolute bioavailability of a drug, when administered by an extravascular route, is usually less than one (i.e., ''F'' <100%). Various physiological factors reduce the availability of drugs prior to their entry into the systemic circulation. Whether a drug is taken with or without food will also affect absorption, other drugs taken concurrently may alter absorption and first-pass metabolism, intestinal motility alters the dissolution of the drug and may affect the degree of chemical degradation of the drug by intestinal microflora. Disease states affecting liver metabolism or gastrointestinal function will also have an effect. |
|||
Other factors may include, but are not limited to: |
|||
* Tính chất vật lý của thuốc ([[kỵ nước]], [[pKa]], [[độ hoà tan]]) |
|||
* Công thức thuốc (tá dược sử dụng, phương pháp bào chế, phóng thích tức thời [[Time release technology|modified release]] – delayed release, extended release, sustained release, etc.) |
|||
* Whether the formulation is administered in a fed or [[fasting|fasted]] state |
|||
* Gastric emptying rate |
|||
* [[Circadian]] differences |
|||
* Interactions with other drugs/foods: |
|||
** Interactions with other drugs (e.g., [[antacid]]s, alcohol, nicotine) |
|||
** Interactions with other foods (e.g., [[grapefruit juice]], [[pomello]], [[cranberry juice]], [[brassica]] vegetables) |
|||
* Transporters: Substrate of [[efflux (microbiology)|efflux]] transporters (e.g. [[P-glycoprotein]]) |
|||
* Health of the [[GI tract]] |
|||
* [[Enzyme]] induction/inhibition by other drugs/foods: |
|||
** Enzyme induction (increased rate of metabolism), e.g., [[Phenytoin]] induces [[CYP1A2]], [[CYP2C9]], [[CYP2C19]], and [[CYP3A4]] |
|||
** [[Enzyme inhibitor|Enzyme inhibition]] (decreased rate of metabolism), e.g., grapefruit juice inhibits CYP3A → higher nifedipine concentrations |
|||
* Individual variation in metabolic differences |
|||
** Age: In general, drugs are metabolized more slowly in fetal, neonatal, and geriatric populations |
|||
** [[Phenotype#Phenotypic variation|Phenotypic differences]], [[enterohepatic circulation]], diet, gender |
|||
* Disease state |
|||
** E.g., [[hepatic]] insufficiency, poor [[renal]] function |
|||
Each of these factors may vary from patient to patient (inter-individual variation), and indeed in the same patient over time (intra-individual variation). In [[clinical trial]]s, inter-individual variation is a critical measurement used to assess the bioavailability differences from patient to patient in order to ensure predictable dosing. |
|||
== Bioavailability of drugs versus dietary supplements == |
|||
In comparison to drugs, there are significant differences in dietary supplements that impact the evaluation of their bioavailability. These differences include the following: the fact that nutritional supplements provide benefits that are variable and often qualitative in nature; the measurement of nutrient absorption lacks the precision; nutritional supplements are consumed for prevention and well-being; nutritional supplements do not exhibit characteristic [[dose-response relationship|dose-response curves]]; and dosing intervals of nutritional supplements, therefore, are not critical in contrast to drug therapy.<ref name="Srinivasan" /> |
|||
In addition, the lack of defined methodology and regulations surrounding the consumption of dietary supplements hinders the application of bioavailability measures in comparison to drugs. In clinical trials with dietary supplements, bioavailability primarily focuses on statistical descriptions of mean or average AUC differences between treatment groups, while often failing to compare or discuss their standard deviations or inter-individual variation. This failure leaves open the question of whether or not an individual in a group is likely to experience the benefits described by the mean-difference comparisons. Further, even if this issue were discussed, it would be difficult to communicate meaning of these inter-subject variances to consumers and/or their physicians. |
|||
== Nutritional science: reliable and universal bioavailability == |
|||
One way to resolve this problem is to define "reliable bioavailability" as positive bioavailability results (an absorption meeting a predefined criteria) that include 84% of the trial subjects and "universal bioavailability" as those that include 98% of the trial subjects. This reliable-universal framework would improve communications with physicians and consumers such that, if it were included on products labels for example, make educated choices as to the benefits of a formulation for them directly. In addition, the reliable-universal framework is similar to the construction of confidence intervals, which statisticians have long offered as one potential solution for dealing with small samples, violations of statistical assumptions or large standard deviations.<ref>{{cite journal |last1=Kagan |first1=Daniel |last2=Madhavi |first2=Doddabele |last3=Bank |first3=Ginny |last4=Lachlan |first4=Kenneth |title='Universal' and 'Reliable' Bioavailability Claims: Criteria That May Increase Physician Confidence in Nutritional Supplements |journal=Natural Medicine Journal |volume=2 |issue=1 |pages=1–5 |year=2010 |url=http://naturalmedicinejournal.net/pdf/NMJ_JAN10_NaPh.pdf}}</ref> |
|||
== Ghi chú == |
== Ghi chú == |
||
Phiên bản lúc 13:56, ngày 20 tháng 3 năm 2013
Trong Dược học, Sinh khả dụng là một địa lượng chỉ tốc độ và mức độ hấp thu dược chất từ một chế phầm bào chế vào tuần hoàn chung một cách nguyên vẹn và đưa đến nơi tác dụng.[1] Được ký hiệu bằng chữ f (hay nếu ở dạng phần trăm là F).
Định nghĩa
Trong Dược học, Sinh khả dụng (hay như trong tiếng Anh là bioavailability (BA)) là đại lượng chỉ tốc độ và mức độ hấp thu dược chất từ một chế phẩm bào chế và tuần hoàn chung một cách nguyên vẹn và đưa đến nơi tác dụng, từ đó tiếp tục được chuyển hoá và thải hồi. Từ định nghĩa, thuốc theo đường tiêm tĩnh mạch có sinh khả dụng là 100%.[2] Tuy vậy, khi thuốc được dùng bằng các cách thức khác nhau (như đường uống) thì sinh khả dụng của thuốc[TH] thường giảm (do hấp thu không hoàn toàn và các giai đoạn đầu của quá trình trao đỗi chất) hay thay đỗi tuỳ thuộc thể trạng bệnh nhân. Sinh khả dụng được xem là một công cụ thiết yếu trong sinh dược học, đây là đại lượng quan trọng để xác định và tính toán liều dùng cho các dạng bào chế không theo đường tĩnh mạch.
Sinh khả dụng tuyệt đối
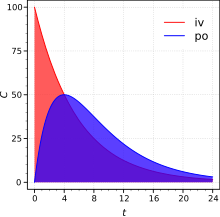
Sinh khả dụng tuyệt đối được xác định khi so sánh sinh khả dụng của thuốc có hoạt tính lưu hành trong hệ tuần hoàn theo cong đường không phải tiêm tĩnh mạch (ví dụ như theo đường uống, đường trực tràng, thẩm thấu qua da, tiêm dưới da hay đạt dưới lưỡi), với sinh khả dụng của cùng dạng thuốc theo đường tiêm tĩnh mạch. Do chỉ có một phần của thuốc hấp thụ theo đường không tiêm tĩnh mạch so với củng dạng thuốc đó khi tiêm tĩnh mạch, nên sự so sánh này phải được thực hiện trên cách liều khác nhau (ví dụ, khảo sát các liều dùng khác nhau hay trên các đối tượng có trọng lượng khác nhau). Từ đó , nồng độ chất hấp thụ sẻ dần được nâng cao bằng cách chia liệu lượng dùng hợp lý.
In pharmacology, in order to determine absolute bioavailability of a drug, a pharmacokinetic study must be done to obtain a plasma drug concentration vs time plot for the drug after both intravenous (iv) and extravascular (non-intravenous, i.e., oral) administration. Sinh khả dụng là liều dùng xác định thể hiện bằng diện tích dưới đường cong (AUC) theo đường không tiêm tĩnh mạch chia cho diện tích dưới đường cong AUC theo đường tiêm tĩnh mạch. Ví dụ, công thức tính F (sinh khả dụng) của một loại thuốc được dùng theo đường uống (po) được cho như sau.
Ghi chú
Xem thêm
Tham khảo
- ^ Shargel, L.; Yu, A.B. (1999). Applied biopharmaceutics & pharmacokinetics (4th ed.). New York: McGraw-Hill. ISBN 0-8385-0278-4Bản mẫu:Pn
- ^ Griffin, J.P. The Textbook of Pharmaceutical Medicine (6th Ed.). New Jersey: BMJ Books. ISBN 978-1-4051-8035-1Bản mẫu:Pn
- ^ Schuppan, D; Molz, KH; Staib, AH; Rietbrock, N (1981). “Bioavailability of theophylline from a sustained-release aminophylline formulation (Euphyllin retard tablets)--plasma levels after single and multiple oral doses”. International journal of clinical pharmacology, therapy, and toxicology. 19 (5): 223–7. PMID 7251238.
Tham khảo thêm
- Malcolm Rowland; Thomas N. Tozer (2010). Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications (ấn bản 4). Philadelphia, PA: Lippincott Williams & Wilkins. ISBN 978-0-7817-5009-7.
- Peter G. Welling; Francis L. S. Tse; Shrikant V. Dighe (1991). Pharmaceutical Bioequivalence. Drugs and the Pharmaceutical Sciences. 48. New York, NY: Marcel Dekker. ISBN 978-0-8247-8484-3.
- Dieter Hauschke; Volker Steinijans; Iris Pigeot (2007). “Metrics to characterize concentration-time profiles in single- and multiple-dose bioequivalence studies”. Bioequivalence Studies in Drug Development: Methods and Applications. Statistics in Practice. Chichester, UK: John Wiley and Sons. tr. 17–36. ISBN 978-0-470-09475-4. Truy cập ngày 21 tháng 4 năm 2011. Đã bỏ qua tham số không rõ
|chapterurl=(trợ giúp) - Shein-Chung Chow; Jen-pei Liu (15 tháng 10 năm 2008). Design and Analysis of Bioavailability and Bioequivalence Studies. Biostatistics Series. 27 (ấn bản 3). Boca Raton, FL: CRC Press. ISBN 978-1-58488-668-6.

